
The state of the art of bispecific antibodies for treating human malignancies | EMBO Molecular Medicine

IJMS | Free Full-Text | LAG-3 as a Potent Target for Novel Anticancer Therapies of a Wide Range of Tumors | HTML
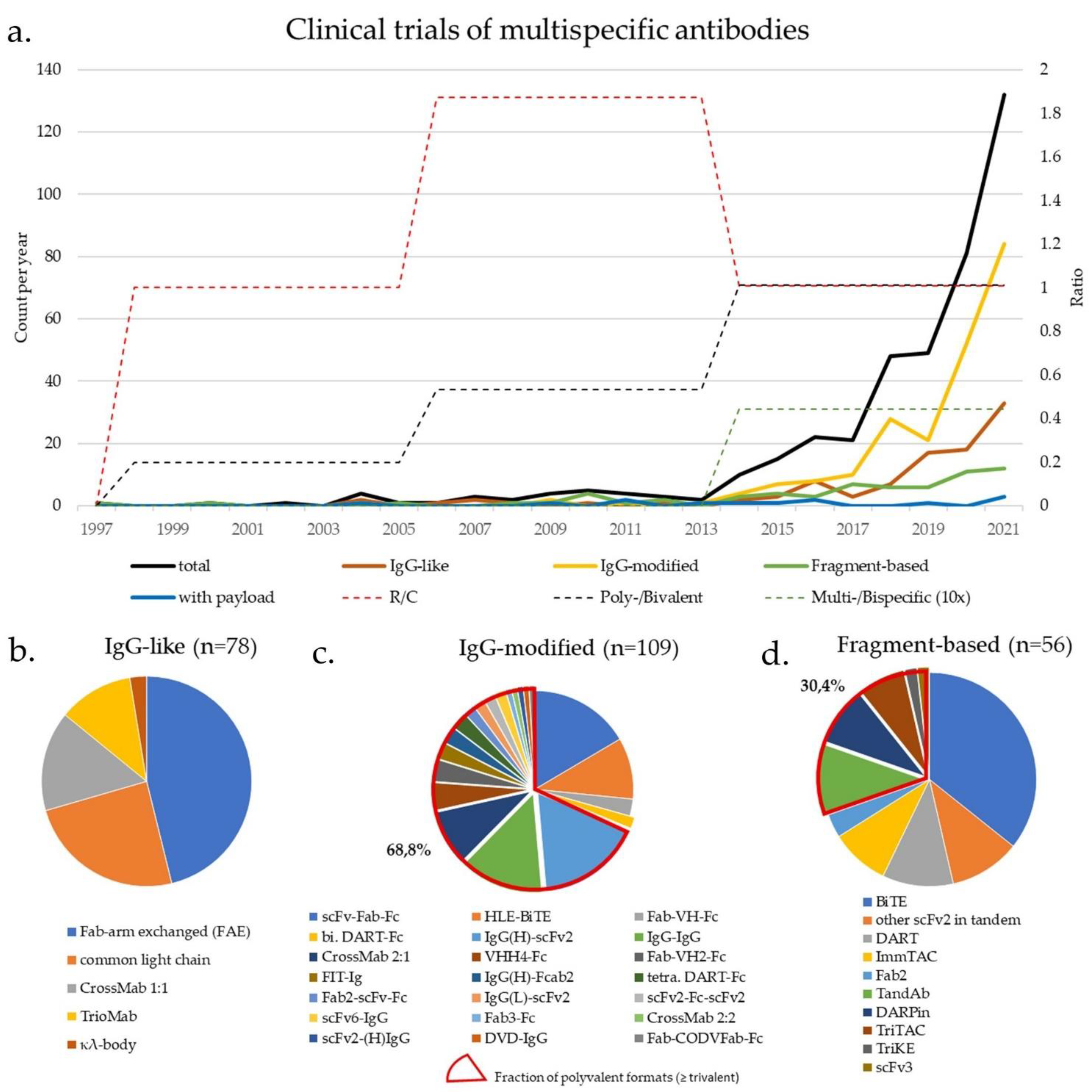
IJMS | Free Full-Text | Principles and Current Clinical Landscape of Multispecific Antibodies against Cancer | HTML

A review of bispecific antibodies and antibody constructs in oncology and clinical challenges - ScienceDirect
A Phase 1, First-in-Human, Open-label, Dose Escalation Study of MGD013, a Bispecific DART® Protein Binding PD-1 and LAG-3 in P

MOAs of 18 bsAbs in Chinese clinical trials. (A) Among the 18 bsAbs,... | Download Scientific Diagram
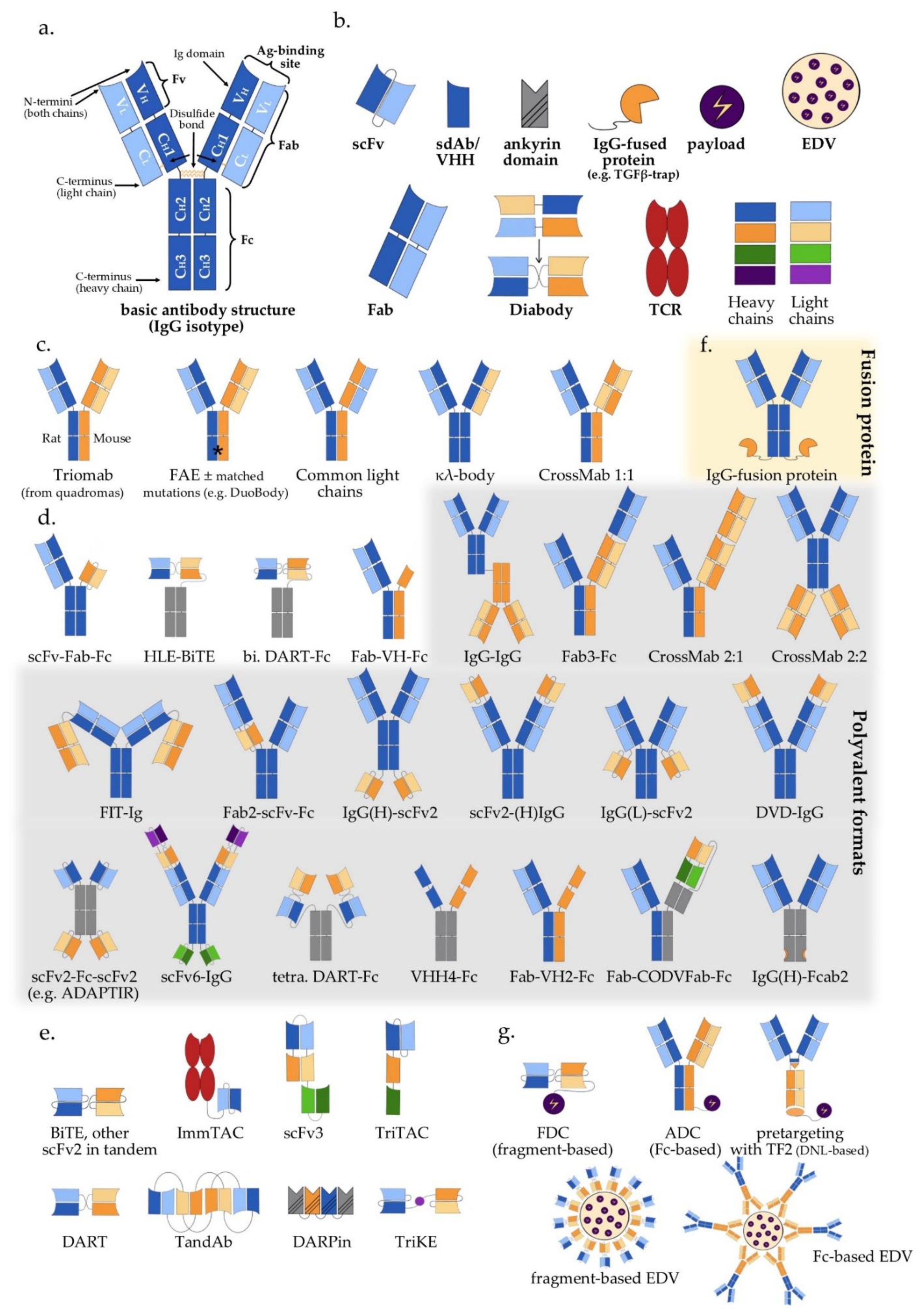
IJMS | Free Full-Text | Principles and Current Clinical Landscape of Multispecific Antibodies against Cancer | HTML

Emerging new therapeutic antibody derivatives for cancer treatment | Signal Transduction and Targeted Therapy

Current Landscape of Immunotherapy Trials Involving the Programmed Cell Death Protein 1/Programmed Death-Ligand 1 Axis in Intrathoracic Tumors - JTO Clinical and Research Reports

The state of the art of bispecific antibodies for treating human malignancies | EMBO Molecular Medicine

Targeted Anti‐Tumor Immunotherapy Using Tumor Infiltrating Cells - Xie - 2021 - Advanced Science - Wiley Online Library

Inhibitory receptors and ligands beyond PD-1, PD-L1 and CTLA-4: breakthroughs or backups | Nature Immunology

Multispecific antibody formats in clinical trials by structure, mode of... | Download Scientific Diagram
A Phase 1, Open-Label Study of MGD013 (Tebotelimab), a Bispecific DART® Molecule Binding PD-1 and LAG-3, in Patients with Relap

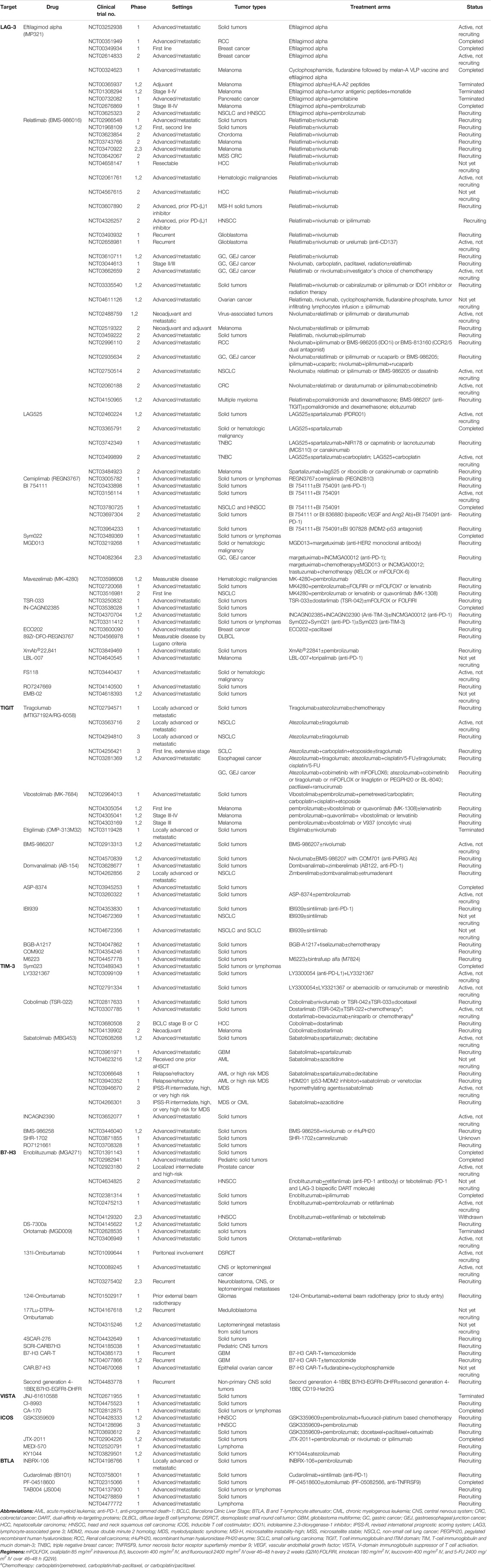

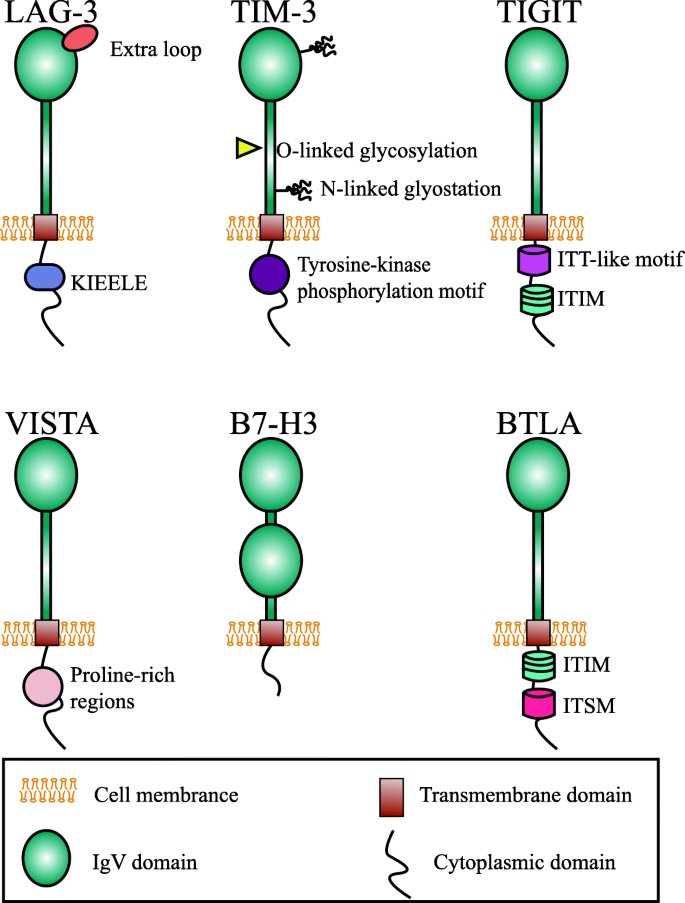


![Top 10 Cholangiocarcinoma Clinical Trials [2022 Studies] | Power Top 10 Cholangiocarcinoma Clinical Trials [2022 Studies] | Power](https://pwr-og-img.withpower.com/Apply%20to%20this%20Phase%202%20clinical%20trial%20treating%20Cholangiocarcinoma,%20Intrahepatic%20Cholangiocarcinoma:%0A%0ADexamethasone%20for%20Cholangiocarcinoma.png?md=1)


